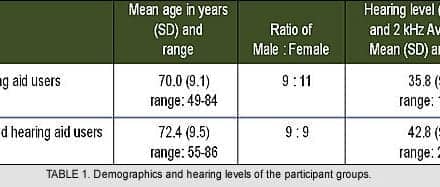
Updated post. Originally published in:
Implants | Winter 2013 Hearing Review Products
The auditory brainstem implant has now benefited over 2,000 people worldwide.
By Steve Otto, MA, and Margaret Winter, MS
In 1979, a patient with neurofibromatosis type 2 (NF2), struggling to save some of her hearing, appealed to her doctors at House Research Institute (HRI) for help, even if it meant trying a surgical treatment never before attempted—an electronic hearing implant directly on her brain.1 The patient’s ear specialists, William House, MD, and William Hitselberger, MD, formulated a revolutionary plan based on pioneering experience with cochlear implants. As a result of this collaboration, this patient soon became auditory brainstem implant (ABI) #1, the first successful auditory brainstem implant user in the world. She continues to use this hearing device daily 34 years later. Worldwide, the ABI now has benefited upwards of 2,000 individuals without functioning auditory nerves, including infants as young as 6 months of age.

Steve Otto, MA, has been Chief Audiologist and Coordinator for the Auditory Brainstem Implant Project since 1989.
ABI #1 and 67 other ABI recipients at HRI became the major group of research participants in a national study that led to US Food and Drug Administration (FDA) approval of the ABI in the year 2000. Presently, the ABI is approved for treating deafness in individuals 12 years of age and older who have lost their hearing from bilateral auditory nerve tumors, a disorder called neurofibromatosis type 2 (NF2).

Margaret Winter, MS, is an audiologist and the former Coordinator of Clinical Services in the research department of the Children’s Auditory Research and Evaluation (CARE) Center at the House Ear Institute (HEI).
The device also can be used to treat deafness in individuals who have lost auditory nerve function from other causes. Because of the loss of the interconnection between the inner ear and the brain, these individuals cannot benefit from powerful hearing aids or cochlear implants. The development of the ABI has proven to be a godsend to many of these individuals.
In 300 adults with NF2 at House Research Institute and House Clinic, we have shown that ABIs provide long-term beneficial hearing sensations and assist people in spoken communication with their associates, friends, and families. This also has been the finding in other ABI centers throughout the world. Approximately 25% of regular users have experienced some degree of word understanding even without lipreading cues. A relatively small percentage of these patients have scored over 80% correct on tests of sentence recognition, suggesting that cochlear implant-like performance is possible.
Originally, the developers of the ABI felt that it would provide beneficial hearing to others besides those with NF2 who did not have functional auditory nerves. Many such adults are now benefiting worldwide. For about 10 years outside the United States, the ABI also has been available to young children born without auditory nerves or cochleae, or whose cochleae are severely ossified from meningitis. There has been, until recently, no approved use of the ABI for this population in the United States.
In the past several years, the Children’s Auditory Research and Evaluation (CARE) Center of the HRI has had the opportunity to program and assess progress of American children who received ABIs in the Verona, Italy, clinic of Professor Vittorio Colletti. Based on our experiences, House Research Institute has received approval by the FDA to begin clinical trials with the ABI in young children. Two other US centers, Massachusetts Eye and Ear Infirmary and the University of North Carolina, also have separate clinical trials.
Inclusion Criteria
The HRI’s clinical trial focuses on safety and early efficacy of the use of this device with young children in a combined effort with House Clinic and Children’s Hospital of Los Angeles. Cochlear Corp’s ABI24 auditory brainstem implant will be used, with Freedom external components. The trial’s objective is to evaluate the surgical and device-related safety issues and to document early outcomes in 10 young children who meet the following inclusion criteria:
- Age 24 to 60 months (with exceptions);
- Bilateral profound sensorineural hearing loss as documented through physiologic and behavioral assessments;
- A diagnosis of one of the following, based on thin-slice magnetic resonance imaging and computed tomography:
- Cochlear aplasia
- Complete cochlear ossification, post meningitis
- Cochlear nerve deficiency (aplasia, hypoplasia)
- Strong family support, to include language proficiency of both parents/legal guardian(s) in the child’s primary mode of communication and primary educational mode of communication (eg, English) or another form of visual support for communication (eg, American Sign Language, Signed English, or Cued Speech);
- Reasonable expectations from parents, to be determined informally by clinicians and psychologist, including:
- Understand benefits and limitations of the ABI
- Understand parental role in rehabilitation, and
- Understand the child may not be successful in developing spoken language as a primary mode of communication or in developing sufficient oral language skills to allow maximum academic progress in an oral environment;
- The child’s parents/legal guardians have proficiency in spoken and written English to enable completion of study-related questionnaires;
- Informed consent from the child’s parents/legal guardian.
Specific to children with a CI:
- Lack of audiological benefit from the implant, at 12 months post-implant (eg, Neural Response Telemetry, evoked cortical responses together with a lack of behavioral responses).
Specific to children without a CI:
- Lack of audiological benefit after a 3-month appropriate amplification trial, except for:
- Post-meningitis subjects with CT evidence of complete ossification, and
- CT evidence of cochlear aplasia or cochlear nerve deficiency.
In addition to these inclusion criteria, specific exclusion criteria include:
- Cognitive and/or developmental delays that would be expected to interfere with the child’s ability to cooperate in testing and/or device programming, in developing speech and oral language, or that would make an implant and subsequent emphasis on aural/oral communication not in the child’s best interest;
- Anomaly/pathology involving the brainstem and/or cortex;
- Retrocochlear pathology resulting from NF2, or other types of cranial nerve/brainstem tumors;
- Coexisting medical conditions that require irradiation of the brainstem and/or auditory cortex; and
- Any medical contraindication, such as cardiopulmonary or renal disease precluding safe administration.
The children enrolled in this clinical trial will be tracked for 3 years. Feasibility will be determined by the children’s ability to detect sounds in the speech range at or better than 50 dB HL in soundfield. An early efficacy study examining outcomes will be conducted for an additional 2 years. Included in the efficacy protocol are speech perception/recognition measures, speech-language evaluations, and adaptive behavior assessments. The speech recognition test protocol is taken from that used in the national Childhood Development after Cochlear Implantation (CDaCI) study,2 which will allow comparison of outcomes among children with ABI, CI, and those with normal hearing.
CANDIDACY EVALUATION
Children will be evaluated to determine candidacy through many of the same procedures and assessments used to determine cochlear implant (CI) candidacy.
Audiologic Evaluation. Qualifying children must have bilateral profound sensorineural hearing loss as determined by both physiologic measures (auditory brainstem response or auditory steady state response, otoacoustic emissions, tympanometry, and acoustic reflexes) and behavioral audiologic assessment using ear-specific, age-appropriate test techniques. They must be determined to have no auditory benefit from appropriately fit amplification. Children who have already received a cochlear implant will be assessed in soundfield to document lack of detection and speech perception benefit from the device.
Medical Evaluation. Anatomy will be examined through use of both CT and MRI scans. Where no cochlea is present and cochlear implantation is impossible, a child may be considered a candidate from an anatomic perspective. Where no auditory nerve is observed or is questionably present, a cochlear implant may be recommended prior to considering an ABI. With children who have already used a cochlear implant without benefit, it is essential that the likelihood of cochlear nerve deficiency be documentable through review of preoperative MRI scans, since there may be other reasons for lack of progress with the cochlear implant. Candidates must be free of any contraindications to the surgical procedure, which will be determined through examination by their pediatrician and the team’s anesthesiologist, neurotologists, and neurosurgeons.
Surgery. The surgical procedure involves a retrosigmoid craniotomy, identification of the VIII cranial nerve and cochlear nucleus, and placement of the ABI device into the lateral recess of the fourth ventricle. The most likely complication of surgery is a cerebrospinal fluid leak, prevention of which is addressed through scrupulous care during closure. A typical hospital stay is 3 to 5 days.
Programming. The levels at which sedated eABR waveforms are visible, without non-auditory side effects, provide the starting levels for initial programming. These levels may or may not provide audibility, so further behavioral mappings are performed in much the same manner as pediatric cochlear implant mappings, using age-appropriate conditioned response techniques. However, tools such as neural response telemetry (NRT) and stapedial reflex measures are not available in ABI mapping, and ABI maps require varying pulse durations and current levels across electrodes to manage non-auditory sensations. Interpolation from only a few measured electrodes would not be acceptable.
Careful observation not only of conditioned responses but also of the child’s behavior is especially important to detect the presence of any non-auditory side effects. In adults with NF2, such non-auditory sensations have been observed on up to approximately 42% of electrodes. It is often possible to reduce the magnitude of such sensations by adjusting stimulus parameters, particularly pulse duration. And mild side effects during testing often are not noticed by ABI recipients during actual use, or disappear entirely with time. In children, where reports are not forthcoming, these effects may be evidenced by general distress, coughing, mild dizziness, gagging, or repeated touching of an area, and it is essential to determine whether these signs are indeed connected to presentation of the stimulus. Significant non-auditory side effects should be avoided and may require turning off an electrode or electrodes. Unlike CI maps where typically most if not all electrodes are active in the map, fewer electrodes may be active in an ABI map if no responses are seen, or if negative responses are seen. It may be possible to add these electrodes later.
Progress assessments are conducted at regular intervals, just as with cochlear implant recipients, and guidance should be provided to parents regarding effective communication strategies and educational placement as the child’s listening and language skills develop.
Language Development. Candidates for this study are in their peak years for optimal language development. Some are expected to have developed age-appropriate visual communication skills; others will have significant language delays through focus on auditory/oral skill development that has not been successful because of their lack of preoperative auditory capacity.
Among the children with ABIs seen in our clinic, most have demonstrated the ability to detect sounds across the speech frequency spectrum within the long-term average speech spectrum (LTASS). Some have developed closed-set speech perception, and several have developed open-set speech perception and have been able to succeed in mainstream educational placements with strong support services.
Considering that outcomes with ABI in this population have been so variable, it is advisable that even as children must receive expert auditory therapy to maximize their benefit from the ABI, they continue to develop their visual communication skills until the trajectory of their progress with the ABI is clear. The expectation is, with the current level of technology, that some children receiving ABIs will always rely strongly on visual language or at least visual supplements to oral language, such as sign language and cued speech. Some children have already demonstrated the ability to use oral language functionally, although they still need to see the speaker’s face, take advantage of captioning, and/or use sign or cued speech for clarification. Existing education programs may not meet the needs of these youngsters without modifications.
Total communication programs that support development of the child’s current communication mode may need to emphasize auditory skill development much more strongly as well. Oral programs where visual language is not used and visual cues are typically minimized may need to include a visual language component, or visual language support, for the child with an ABI.
It is essential to remember that, as the safety and efficacy of this technology are explored in young children, the primary goal should be the development of a language system through which the children are able to succeed communicatively and educationally. Determination of the best strategies to achieve this goal with children who are provided access to sound through the ABI will likely require flexibility, creativity, and special dedication by professionals and parents alike.
REFERENCES
-
Hitselberger WE, House WF, Edgerton BJ, Whitaker S. Cochlear nucleus implants. Otolaryngol Head Neck Surg. 1984;92:52-4.
-
Eisenberg LS, Johnson KC, Martinez AS, et al; CDaCI Investigative Team. Speech recognition at 1-year follow-up in the childhood development after cochlear implantation study: methods and preliminary findings. Audiol Neurootol. 2006;11(4):259-268.