Most of us accept Newton’s idea of synergy: The whole is greater than the sum of its parts. This concept also describes the role and shared efforts that sophisticated electromechanical components play in the success and long-term durability of hearing instruments. Not only is the whole (the hearing device) greater than the sum of its parts (components and sub-components), each electromechanical component—precisely because of its relation to the other parts—is itself enhanced and evolved from the product development and quality control (QC) process. Further, each electromechanical part relies on other electromechanical parts to function properly. If one part malfunctions, in many cases, there can be serious consequences in terms of performance and function of the hearing device.
Hearing instruments endure a fairly extreme environment. Although relative humidity (30%-60% RH) and temperature levels (80°-90°F)1,2 within a typical hearing aid application are not overly severe, these conditions are certainly more rigorous compared to what typical electronic devices face on a constant basis. The ear canal and head also create an environment that can include moisture (eg, rain), debris, cerumen, and corrosive salt from the wearer’s sweat.3
Furthermore, wearers can be quite rough; improper use of button or volume control knobs are common. This can cause breakage and functional failures in the essential electromechanical components that reside in the hearing aid.
Dealing with these types of demands— in addition to the constant drive to miniaturize hearing instruments—has been the key challenge of electromechanical component producers for decades. To meet these challenges, component manufacturers have set up elaborate quality assurance, testing, and risk departments. Sonion A/S in Denmark created its own integrated quality assurance and risk assessment program in 1992, and was the first acoustical electromechanical company to be ISO 9001 approved in 1994. ISO 9001 is a quality assurance model made up of 20 sets of quality system requirements.
Through the years, Sonion has produced more than 80 million electromechanical components and is one of the largest producers of miniature electromechanical components worldwide. The company’s production facilities in Poland are both LEAN certified and “Six Sigma.” Six Sigma is one of the highest quality measures possible in production. A “sigma level” is the number of standard deviations that can fit between the “mean” and the “nearest specification.” This allows the translation of a sigma level into a numerical probability. Thus, sigma is a quality measure used to compare the capability of a process to meet a specification; it can be used as an indicator that correlates directly to a defect rate. The lower the sigma level, the higher the defect rate. A sigma of six is the lowest possible level. Most companies today operate between the 3- and 4-sigma levels.

In-House Quality and Reliability Testing
Quality and reliability testing requires large-scale investments in test equipment and personnel if it is to be carried out in-house. However, having an in-house quality control department can provide numerous quality assurance benefits, including:
- The test processes are stable and repeatable, which means you can trust the test documentation.
- State-of-the-art test equipment is often built in-house to be able to live up to superior accuracy requirements (Figure 1).
- It allows for adjusting test conditions to market standards and develops better product-specific tests. In-house testing facilities also give the possibility to perform customer-specific tests.
- Test results are actively fed back to R&D for build-in quality already in the earliest phase of the development process.
Integrated Quality Assurance
The hearing instrument market demands ever-smaller components with more functionality. To fulfill this requirement, it is necessary to push the limits of what is physically possible without compromising quality.

FIGURE 2. The integrated quality assurance process.
Figure 2 shows Sonion’s Integrated Quality Assurance approach which is designed to ensure a high quality level from the early development phase to running production. For example, one of our flagship products, PJ 185, is run through a quality inspection 28 times during its production life. Both mechanical and electrical parameters are checked—from the inspection of the raw materials upon arrival, to the operating torque of switches, to the synchronization of switch moment and electrical connections.
A systems approach to quality control. Experience also shows that design changes are much easier to implement in the beginning of the development phase than later in the process. If, for example, the automated production processes are not considered from the beginning, it is likely that a redesign will be needed when the final electromechanical product is about to go into production. This is both costly and time consuming. Therefore, a systematic approach has been created to integrate quality in the early phases of the development project.
Risk assessment during development. A systematic Failure Mode and Effect Analysis (FMEA) program is initiated early in the development phase in critical to high levels of quality assurance. The FMEA focuses on the risks associated with the functional design and production-related issues (Figure 3).

FIGURE 3. Table used in FMEA analysis.
A key task in the FMEA process is to estimate the likelihood of a failure to occur. However, this is a very subjective estimate that contains a lot of uncertainty. To overcome this uncertainty, engineers decided that every function, process, or component that is classified as “critical” in the FMEA will be further analyzed even though there was a minute likelihood of the occurrence.
Material qualification. Any engineer will tell you that the robustness of the individual sub-parts determines the quality of the finished component. A good quality assurance program performs material tests on all sub-parts to verify their properties in the earliest phases of development. Requirements are specified and systematic testing is carried out methodically. This includes tests for wear resistance, electrical conductivity, mechanical loads, chemical composition, etc.
Close cooperation with suppliers is essential. Suppliers are qualified according to precise requirements and are re-qualified within regular intervals. This ensures that the required capability of the supplier is maintained and further developed.
Sonion Quality Certificate (SQC). Sonion issues a SQC when a new component has successfully passed a specified test protocol. Introduced 15 years ago, the SQC testing concept has been a key success factor in establishing Sonion as a supplier of high quality components. The testing is done prior to volume production and release of the product for sale. (The SQC Report is available to Sonion’s customers upon request.)
A large number of tests are included in the SQC, including environmental exposures, mechanical tests, and wears tests. Some tests are performed for all products, and some tests are specific to the individual product type.
To keep a high accuracy in temperature and humidity control during environmental exposure, Feutron climactic chambers are used, and these are calibrated at regular intervals.
Due to the small dimensions and close tolerances required, mechanical tests and wear tests are carried out using tools that have been customized by our own tool shop. Mechanical tests include toggle/knob activation torque and force, retention force of terminals and knobs, rotational torque and mechanical feeling performed by certified operators.
Electrical tests include resistance value, pulse characteristics, contact resistance, insulation resistance and parasitic capacity. Audible sound characterization—using ultra-low-noise mechanical test equipment—is also performed to identify the perceived product sound quality.
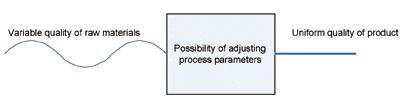
FIGURE 4. Adjusting process parameters.
Qualification of production equipment. To ensure a high and consistent quality level, Sonion qualifies the production equipment prior to volume production (Figure 4). Qualification is done in a Pilot Production Unit and is a combination of qualifying the processes and the equipment. The critical functions and processes are identified, and these are systematically tested to document the reliability of the production process. All tests are documented in order to be able to repeat and compare them with the original tests when changing functions or processes.
Continuous production quality assurance. To ensure seamless transition from pilot production in Denmark, to high volume production at the Polish production facilities, Sonion has established a number of quality assurance activities:
Quality control and receiving control during production for sub-parts. A large number of sub-parts are used in the final hearing instrument component product. All sub-parts used in the final product need to pass the incoming inspection. A specific random test verifies that the part fulfills the pre-determined requirements.
During the production of these sub-parts, Inline Process Control (IPC) is used by continuously measuring the parameters which are defined as critical. Key parameters of the product are tested continuously during the production process.
Additionally, to ensure the production test equipment is according to specifications, it is calibrated against the Quality Assurance test equipment. Production tests are repeated on this equipment (Figure 5).
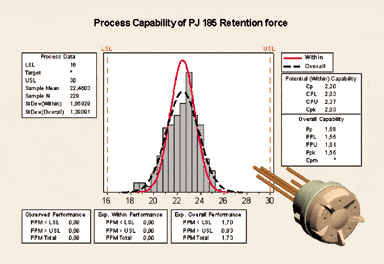
FIGURE 5. Process capability of an electromechanical component (PJ 185 volume control).
Re-qualification change management. Whenever a component has a material or process change, a re-qualification test is conducted to ensure the quality level is maintained—or increased. All samples tested are kept for a minimum of 5 years.
Conclusion
The world is unpredictable, and the environment in which hearing aids operate (eg, on the ear or within the ear canal) can be extreme. High quality products that are continuously developed and refined through exacting test methods, as well as strict manufacturing processes, help to standardize performance of hearing instruments and eliminate the negative aspects of unpredictability.
High product quality benefits everyone involved in the hearing health care process. Hearing instrument manufacturers save on service and repair expenses, while maximizing production yield. Additionally, hearing instrument development time is made more predictable because problems with the application of the electromechanical component are reduced. For dispensing professionals, fewer breakdowns in hearing instrument components means fewer headaches in the form of patients dealing with a malfunctioning device or waiting for a hearing aid repair/replacement. And, of course, a more dependable, reliable, and longer-lasting hearing instrument helps contribute to customer satisfaction with the device.
Quality control and assurance procedures have been promoted throughout the years, and hearing industry components manufacturers have a reputation of focusing on quality and reliability. Sonion views this facet as one of the main reasons for its world-leading position within the electromechanical components for the hearing instrument market.
| This article was submitted to HR by Morten Wagner, BSEE, product manager for Electromechanical Components, and Søren Lyngsø-Petersen, PhD, director of production engineering and quality-EMC at Sonion A/S, Roskilde, Denmark. Wagner has published several reports on behalf of Delta, the Danish Research Center for Applied Electronics, and Petersen has 15 years experience in pharmaceutical industry quality control and has written several books on the topic of validation and testing in the health care field. Correspondence can be addressed to HR or Morten Wagner, Sonion Roskilde A/S, Byleddet 12-14, DK-4000, Roskilde, Denmark; email: [email protected]. |
References
1. Agnew J. Temperature and relative humidity inside an ITE hearing instrument. The Hearing Review. 1999;6(8):28-34,73.
2. Bailey JW, Valente M. Measurements of relative humidity and temperature in hearing aids. Hear Jour. 1996;49(10):59-63.
3. Agnew J. Towards a better understanding of cerumen. The Hearing Review. 1994;1(4):16-20.